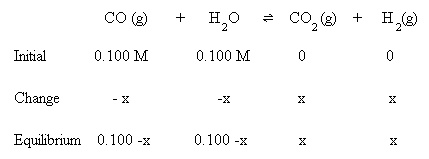| Recognizing Equilibrium | Thermodynamics and Equilibrium | Equilibrium Constant (I) | Equilibrium Constant (II) |
| Predicting the Direction of a Reaction (I) | Predicting the Direction of a Reaction (II) |
| Predicting the Direction of a Reaction (I) | Predicting the Direction of a Reaction (II) |
Equilibrium Constant (I)
|
Lesson:
In this lesson, the instructor will introduce the law of chemical equilibrium, as well as the equilibrium constant. After a thorough understanding of the two topics, the instructor will give a brief introduction to the ICE table. A very simple example is to be made, since this will be a fresh concept for the students. Consolidation:
Exit slips will be used as a consolidation. It is important that the students understand this concept, as this is present throughout this whole unit. Students will write down 1 thing that they do NOT understand, and 1 thing that they learnt through this lesson. |
***If you wish to flip the classroom for this lesson (i.e. focusing on problem solving in class, and assigning the "lesson" for homework), I recommend giving this video for the students to watch)***
For students who would want extra information, this page is a good web page for this particular topic. Even if you aren't flipping the classroom, the video and the webpage is excellent for extra support. |
LAB 1:
In this lab, which is to be prepared following this lesson, students will be able to measure the equilibrium constant for a simple reaction.
In this lab, which is to be prepared following this lesson, students will be able to measure the equilibrium constant for a simple reaction.
Visiting this site will also aid with the preparation of this lab.